Embolisation of Symptomatic Pelvic Veins in Women Presenting with Non-saphenous Varicose Veins of Pelvic Origine – Three year Follow-up

Embolisation of Symptomatic Pelvic Veins in Women Presenting with Non-saphenous Varicose Veins of Pelvic Origine – Three year Follow-up
Abstract
Aim. To evaluate the clinical results of embolisation of symptomatic, incompetent pelvic veins in women presenting with perineal veins.
Patients and methods. Twenty-four women presenting with non-saphenous perineal varicose veins and who experienced pelvic vein syndrome were treated. Symptoms was scored on a visual analogue scale assessing dyspareunia, pelvic and lower limb pain. Lower limb varices were investigated by duplex ultrasonography. Pelvic veins were studied by pelvic vein angiography with simultaneous embolisation of incompetent veins. Ovarian and internal iliac veins were systematically embolised when incompetent. Follow-up assessment of symptoms and varices was carried out at 1, 2 and 3 years.
Results. All patients presented with perineal veins, 2 with sciatic vein incompetence and 2 with a perforator of the thigh or buttock. Pelvic venous angiography was performed via right femoral access in 87% of the cases and confirmed the presence of incompetent ovarian and internal iliac veins. The mean number of coils used per vein was 6 and all were successfully embolised. No serious complications were encountered. The mean clinical improvement score was 80%, 77%, 80% and 76% at respectively 45 days, 1, 2 and 3-year follow-up.
Conclusion. In women of reproductive age, non-saphenous varicose veins associated with pelvic venous incompetence(PVI) should undergo pelvic vein investigation. In this clinical series we achieved a satisfactory improvement in symptoms after 3 years following treatment of incompetent pelvic veins.
Introduction
In women, clinical symptoms of pelvic vein incompetence(PVI) have been recognised for years.1 They were often considered an inevitable part of the chronic pelvic pain that young multiparous women had to suffer. The first surgical treatments were achieved by transperitoneal ligation and division of ovarian veins, but this is a very invasive treatment and is no longer carried out. Modern radiological investigation of pelvic veins has made it possible better to understand, pelvic vein anatomy and anastomoses between abdominal and lower limb veins2 as well as the pathological role of PVI in clinical pelvic symptomatology. Duplex ultrasound studies have shown that varicose veins do not always originate from saphenous systems,3,4 especially in the case of recurrent varices.5,6 Varicose veins of the lower limb may depend solely on pelvic vein incompetence. Evaluation of pelvic symptoms may indicate the presence of a pelvic aetiology of varicose veins. In a previous study,7 a significant correlation between radiological PVI and pelvic clinical symptoms has been demonstrated.
The aim of this series was to use evaluation of symptoms in women of reproductive age to select those where pelvic venous embolisation was indicated amongst those presenting with non-saphenous varices and to evaluate the outcome at three years.
Materials and Methods
Patients
Between 2000 and 2002 amongst 895 female patients assessed in a phlebological practice, 24 non-menopausal women were selected who presented with nonsaphenous varices of pelvic origin and clinical symptoms of PVI. The first criteria for selection was the presence of non-saphenous varices of pelvic origin : varicose veins at buttock level, perineal or vulval varicose veins, or varicose veins situated at the popliteal fossa fed by PVI via the sciatic nerve vein. Duplex ultrasound examination was carried out in the standing position (Hitachi Katana 5, 7.5 MHz transducer; Ecoscan, Les Ulis, France). Patients who had previously undergone varicose vein surgery were assessed to ensure that no varicose veins originated from the previously treated sapheno-femoral junction. The deep system was also studied to exclude patients with ‘nutcracker syndrome’(mesoaortic compression of the left renal vein). Patients whose varicose veins originated principally from connections with the femoral vein were excluded.
The second selection criteria depended on the extent of symptoms of PVI. Among the various symptoms of PVI, as described by Taylor,1 (premenstrual or menstrual pelvic pains, dyspareunia, vulval congestion, dysuria, bladder irritability and urgency, digestive disorders, medical history of vulval varicose veins during deliveries, and haemorrhoids), only 3 particularly clear symptoms were chosen to be evaluated. These were selected based on the findings of a previous study,7 in which a significant correlation between radiological PVI and these three symptoms was demonstrated. Symptoms were assessed by 3 visual analogue scales (VAS) graduated from 0 to 10 (0 was no pain and 10 the maximum pain) completed by the patient during consultations :
- VAS for pelvic menstrual pain occurring specifi- cally before or during menstruation (0-10)
- VAS for pain on the varicose vein tracks occurring specifically before or during menstruation (0-10)
- VAS for dyspareunia occurring specifically during or after intercourse (0-10)
The scores obtained were added to produce a global symptom score ranging from 0 to 30. Patients with no pain or a score below6were excluded.Non-menopausal women with a score above 6 were chosen for inclusion. We did not assess the presence of incompetent pelvic veins by transvaginal ultrasonography since this has been found to be an unreliable technique.8 Before the procedure, all patients gave informed written consent and were asked to return for clinical examination and symptom assessment at 45 days, and at 1, 2 and 3 years after the intervention. One day prior to the planned varicose vein surgery, all patients underwent pelvic venous angiography with embolisation of incompetent pelvic veins.
Pelvic vein angiography and embolisation
Pelvic vein angiography was carried out via a femoral vein canulation with a 4 or 5 French introducer under local anaesthesia and neuroleptanalgesia. Percutaneous catheterisation of the venous segments was carried out with a Sindewinder catheter (Cordis SA, Johnson&Johnson Company, Issy les Moulineaux, France), and a hydrophilic guide support (Te´rumo, Guyancourt, France) with a flexible tip. The veins were selectively catheterised in the following order: the left renal vein, the left ovarian vein, the right ovarian vein, the left internal iliac vein, the left external iliac vein, the right internal iliac vein and the right external iliac vein.
Venous opacification was first carried out at rest and then under pressure (Valsalva manoeuvre), with a hydro-soluble, hypo-osmolar contrast compound at a 270 mg/ml concentration (Visipaque 270, Amersham-Health, Pantin, France), at a dose of 9 to 12 ml, at a 3 to 4 ml/sec flow rate.
Diagnostic and therapeutic procedures were carried out simultaneously. Each ovarian or internal iliac vein with significant reflux was embolised at the same time as the selective catheterisation, and prior to investigation of the next part of the venous system. The pathological reflux criteria that were taken into account to carry out embolisation were :
- Voluminous ovarian varicocele (fed by ovarian vein reflux, evident on injection of the left renal vein).
- Varicose reflux toward the ipsi- or contro-lateral proximal thigh.
- Reflux toward perineal and vulvar veins.
- Reflux toward the ipsi-lateral sciatic vein.
Embolisation was carried out with small metallic coils (Cook, Bloomington, USA) with a 0.03800 calibre, a length of 3 to 8 cm, and a diameter of 3 to 10 mm. Coil size was selected according the calibre and the morphology of the affected venous segment. In an ovarian vein, coils were placed at the lower level of the sacro-iliac joint and at the level of the L5 and L4 vertebrae, to adapt to possible upwardly situated collaterals, until complete occlusion and absence of reflux were obtained under abdominal pressure. In an internal iliac or a sciatic vein, highly selective catheterization was performed up to the origin of the incompetent vein to embolise it. No glue, foam, or sclerosing product was used.
Surgery
After pre-operative ultrasound marking of varices(Esaote AU 530, 10 MHz transducer), phlebectomies were carried out under loco-regional anaesthesia with a femoral block. The number of phlebectomy incisions, which corresponds to the extent of the varices, was also noted. Post operative compression was achieved by two superimposed class II French stockings (2 x 20 = 40 mm Hg) for two days and then by one stocking for 2 weeks.
Clinical outcome
Outcome was assessed on clinical freedom varices and symptoms of pelvic pain. Assessment was performed by the same investigator, after one month to evaluate symptoms during the following menstrual periods. At one, two and three years, the same assessment was repeated. During these consultations the same 3 VAS was completed and the global symptom score was calculated. Patients who were unable to come for a consultation, were interviewed on the phone.
The clinical assessment of varices was divided into 4 categories :
- Type 1 (Excellent result): Absence of varicose veins(equivalent to class C0, C1 of the CEAP classification)
- Type 2 (Good result): Presence of only very few new varices
- Type 3 (Poor results): Presence of new varices, but fewer than previously
- Type 4 (Bad results): Presence of the same amount of varicose veins as before treatment.
Patients who were unable to attend for clinical evaluation at the 3-year follow-up were asked to answer a questionnaire constructed with the same definitions as above.
Statistics
Scores were measured on 5 occasions: before embolisation, at 45-days, 1, 2 and 3-year follow-up. A statistical analysis was performed with SAS v8 software. The score and time at follow-up assessed by analysis of variance. Means were compared two by two with the Scheffe method. Influence of preoperative dyspareunia and, impact of the impossibility to explore the right ovarian vein on the final clinical score, were studied by variance analysis using GLM SAS procedure completed by Scheffe method when the global test was significant.
The comparison of clinical results with the type of recurrences was performed by one factor analysis of variance. Due to the small number of patients in each group concerning varicose veins type 1 and 2 and similarly type 3 and 4 were gathered respectively into 2 types ‘‘good’’ or ‘‘bad’’ results. Statistical significance was accepted for a P value of less than 0.05.
Results
The mean age of patients was 41.5 years (range 31-50). The mean number of pregnancies was 2.5 per patient (range 1-4). When a patient had previously undergone surgery on one side only, embolisation was performed on the corresponding side. Haemorrhoidal pathology was found in 63% of the cases, and voluminous vulvar varicose veins were present at delivery in 47% of the cases. For only 3 patients with history of deep venous thrombosis, embolisation was performed on the same side. Only 2 patients were of CEAP clinic class C3; the remaining 22 were CEAP class C1 or C2. Of the 24 patients, 10 had been previously undergone surgical treatment for varicose veins, with 3 left, 1 right, 4 bilateral great saphenous vein stripping procedures and 2 perforator ligations at the buttocks; 3 other patients had been operated on twice previously and had been treated by phlebectomy alone. The distribution of varicose veins is reported on the Table 1. The number of phlebectomy incisions related to the number of varicose veins was 34 for the left limb (range 10-90) and 32 for the right limb (range 12-65).
Results of embolisation
Pelvic venous angiography was performed by right femoral access for 20 cases, left femoral access for 3 cases, and bilaterally for 1 case. All patients selected on clinical criteria for inclusion in this series were found to have radiologically demonstrated PVI. The distribution of the embolised pelvic veins is reported in the Table 2. In one case sciatic venous reflux was fed by both internal iliac veins (left and right). All these incompetent veins were successfully embolised.
Attempts at selective catheterisation failed in 14 cases for the right ovarian vein, in one case for the left internal iliac vein, and in one case for the right internal iliac vein. During radiological investigation, the absence of flow to the right side of the vena cava suggested a competent right ovarian vein of small calibre. On average, the number of used coils was 7.3 per ovarian vein (range 3-11) and 5.6 per internal iliac vein (range 3-9).
The only immediate complication was the migration of a 3 mm coil, just after deployment in a branch of the left internal iliac vein; it migrated into the lower lobar branch of the left pulmonary artery with no clinical consequences.
Symptoms
All patients were re-evaluated at 45-days, 1, 2 and 3 years, except for 2 patients who were lost from the 3-year follow-up. Two other patients were unable to come for the 3-year follow-up and but were interviewed by telephone.
At 45-days, pain assessed by VAS showed a significant amelioration in symptoms. Pelvic pain was evaluated at a mean score of 1 (range 0-4), dyspareunia at 0.3 (range 0-4) and pain on the former perineal or buttock veins were at a mean score of 1.7 (range 0-7). The difference was statistically significant between each preoperative and postoperative sign (P <.001). (Fig. 1.). No difference appears in the follow-up of each postoperative clinical sign at 45 days, 1, 2 and 3 years.
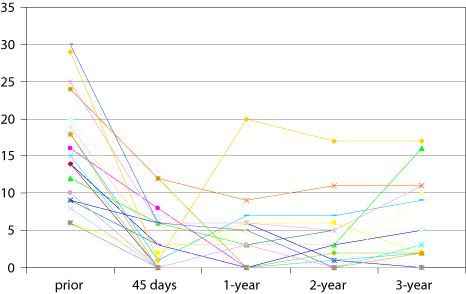
After-45 days, the clinical score had decreased from 15.3 to 3.1, with maintenance of the improvement at the 3 years follow-up.
At 2-year follow-up, 67% of patients had improved symptoms by more than 80%.
Only one patient showed no improvement, and had a previous score of 6 before embolization.
At 3-year follow-up, 68% of patients had improved symptoms by more than 80%.
Improvement of the global symptom score was respectively 77%, 80% and 76% at one, 2- and 3-year follow-up. One patient (with only a 31% clinical improvement) was considered a failure as she presented with a painful pelvic recurrence four months after embolisation (Fig. 2). This patient had successfully undergone left ovarian vein embolisation but it had not been possible to explore her right ovarian vein. One year later, angiography showed that the left ovarian vein was totally obstructed; no pelvic venous incompetence could be detected in the veins that could be studied, but the right ovarian vein still could not be demonstrated. Among the 9 patients who had no symptoms at the 3-year follow-up, 4 no longer had menstruation (menopausal patient or hormonal intrauterine device). In our study, we did not find any correlation between a low clinical score and our inability to investigate the right ovarian vein.
Varicose veins
Among the 22 patients re-evaluated 3-years following treatment, only 10 presented with a type 1 recurrence (no varices). Among the patients who presented with new varices, 7 presented with a type 2, 3 a type 3, and 2 a type 4 recurrence (bad results). Two patients presented with other varices in another part of the limb (small saphenous vein or great saphenous vein). Four patients who could not come to the clinical evaluation at 3-year follow-up answered the questionnaire. One had a clinical result type 1 and the other a type 4.
Due to the small number of patients in the 4 types of results no significant difference can be demonstrated concerning the final clinical score. After gathering these 4 groups into only 2 groups: good results (type 1 + 2) bad results (type 3 + 4) a significant association was demonstrated between the patients with a poor symptom score and varicose vein recurrence (bad results) (P <.001).
Dyspareunia
Dyspareunia noted at the preoperative evaluation was not significantly associated with poor clinical results (P <.079). The slight increase of the score at 3-year follow-up was partly due to the small increase of the level of dyspareunia.
Discussion
The outcome in our series of patients shows that embolisation as treatment for PVI gives satisfactory clinical results at 3-years follow-up. We used pelvic vein angiography for our diagnostic technique which was combined with treatment of affected veins. Others have used duplex ultrasound investigation, but this is controversial. Ovarian vein duplex scanning has been unsuccessfully attempted in some series.8 For others this provides an adequate evaluation in a large percentage of cases.9-12 One authors reports that PVI can be easily detected by putting a CW Doppler probe on vulval varicose veins and on ‘‘point P’’ and ‘‘point I’’.13 Duplex ultrasonography remains essential to eliminate a ‘‘nutcracker syndrome’’.14
In our study, 14 right ovarian veins (58%) could not be catheterised. It seems difficult to catheterise the right ovarian vein from femoral vein access, which is why Leal Monedero15 advises the use of the brachial vein to access the upper vena cava (only 18% of right ovarian vein catheterisations were not successful). With a left femoral vein puncture, Maleux16 reports only 2% of right ovarian vein catheterisation failures. Some authors advise a double right and left femoral access with ovarian and internal iliac investigation carried out as two stages, and others17 prefer the transbrachial approach.
Complications of embolisation reported in medical literature are rare. Accidental embolisation of glue fragments,16 perforations of the ovarian vein with extravasation of contrast, transient cardiac arrhythmia, and coil embolism8 have been described, particularly from the internal iliac vein level.18 Our results compare with those of other series. Unfortunately, there are very few published studies with results at three years follow-up (Table 3).
Patient follow-up beyond a few years is difficult to carry out, since pelvic pain is often an intricate pathology. Changes in oral contraceptive treatment, intra-uterine hormone-liberating devices, or premenopausal hormonal substitution can lessen or even eliminate a painful relapse. Moreover, symptoms tend to decrease spontaneously with the passage of time without any treatment. On the other hand, a new pregnancy can generate a painful relapse. The results concerning varicose vein recurrences are also difficult to evaluate. Even if there appears to be a correlation between symptom recurrence and varicose vein recurrence it is difficult to claim that embolisation can prevent varicose vein recurrence. At present, no studies have been able to clarify this. This may in part explain why indications for embolisation are more questionable in post-menopausal women, who usually have few symptoms which could be improved by treatment.
In pre-menopausalwomen, non-saphenous varicose veins of the upper thigh associated with symptoms of PVI suggest that the pelvic veins should be investigated as a possible source of symptoms. Ovarian veins and internal iliac vein tributaries are often simultaneously incompetent, justifyingmultiple embolisation to address all possible sources of varices and symptoms.
Acknowledgements
We thanks Ms Marie Jose´ Schantz Fleurent and Ms Dominique Vala for their technical help in editing the text for the English language.
REFERENCES
- TAYLOR Jr HC. Vascular congestion and hyperhemia: the effect on function in the female reproductive organs. Part I. Physiological basis and history of the concept. Am J Obstet Gynecol 1949;57: 211e230.
- CASTRO CASTRO J, ZUBICOA EZPELETA S, CADENA CORRALES J, CARRION OTERO EO, LEAL MONEDERO J. Anatomia del sistema venoso de los miembros inferiors y de la pelvis. In: LEAL MONEDERO J, ed. Insuficiencia venosa cronica de la pelvis y de los miembros inferiores. Madrid: Mosby; 1997:1e11.
- LABROPOULOS N, TIONGSON J, PRYOR L, TASSIOPOULOS AK, KANG SS, MANSOUR A et al. Nonsaphenous superficial vein reflux. J Vasc Surg 2001;34:872e877.
- JIANG P, VAN RIJ AM, CHRISTIE RA, HILL GB, THOMSON IA. Nonsaphenous venous reflux in the groin in patients with varicose veins. Eur J Vasc Endovasc Surg 2001;21:550e557.
- GIANNOUKAS AD, DACIE JE, LUMLEY JS. Recurrent varicose veins of both lower limbs due to bilateral ovarian vein incompetence. Ann Vasc Surg 2000;14:397e400.
- LEAL MONEDERO J, ZUBICOA EZPELETA S, CASTRO CASTRO J, CALDERON ORTIZ M, SELLERS FERNANDEZ G. Embolization treatment of recurrent varices of pelvic origin. Phlebology 2006;21:3e11.
- CRETON D, HENNEQUIN L. Insuffisance veineuse pelvienne chez la femme présentant des varices pe´rine´ales. corre´lation anatomoclinique, traitement par embolisation et re´sultats (31 cas). Phle´bologie 2003;56:257e264.
- CORDTS PR, ECLAVEA A, BUCKLEY PJ, DE MAIORIBUS CA, COCKERILL ML, YEAGER TD. Pelvic congestion syndrome: early clinical results after transcatheter ovarian vein embolization. J Vasc Surg 1998;28:862e868.
- CAPASSO P, SIMONS C, TROTTEUR G, DONDELINGER RF, HENROTEAUX D, GASPARD U. Treatment of symptomatic pelvic varices by ovarian vein embolization. Cardiovasc Intervent Radiol 1997;20:107e111.
- GIACHETTO C, COTRONEO GB, MARINCOLO F, CAMMISULI F, CARUSO G, CATIZONE F. Ovarian varicocele: ultrasonic and phlebobraphic evaluation. J Clin Ultrasound 1990;18:551e555.
- PIERI A, VANNUZZI A, NICOLUCCI A, LUDOVICI M, CAILLARD P, VIN F. L’e´cho-doppler couleur transvaginal dans l’e´tude des reflux pelviens et de la varicoce`le fe´minine. Phle´bologie 1999;52: 45e51.
- PARK SJ, LIM JW, KO YT, LEE DH, YOON Y, OH JH et al. Diagnosis of pelvic congestion syndrome using transabdominal and transvaginal sonography. Am J Roentgenol 2004;182:683e688.
- FRANCHESCHI C, BAHNINI A. Treatment of lower extremity venous incompetence due to pelvic leak points in women. Ann Vasc Surg 2005;19:284e288.
- SCULTETUS AH, VILLAVICENCIO JL, GILLESPIE DL. The nutcracker syndrome: its role in the pelvic venous disorders. J Vasc Surg 2001;34:812e819.
- LEAL MONEDERO J. La pathologie veineuse pelvienne. Traitement embolisant. Phle´bologie 1999;52:299e310.
- MALEUX G, STOCKX L, WILMS G, MARCHAL G. Ovarian vein embolization for the treatment of pelvic congestion syndrome: longterm technical and clinical results. J Vasc Interv Radiol 2000;11: 859e864.
- GARRETT JP, WETTON N, TYRREL MR. Ovarian vein embolisation as an adjunct in the treatment of vulval and atypical leg varicosities. Phlebology 2002;17:3e9.
- VENBRUX AC, CHANG AH, KIM HS, MONTAGUE BJ, HEBERT JB, AREPALLY A et al. Pelvic congestion syndrome (pelvic venous incompetence): impact of ovarian and internal iliac vein embolotherapy on menstrual cycle and chronic pelvic pain. J Vasc Interv Radiol 2002;13:171e178.
- PIERI S, AGRESTI P, MORUCCI M, DE’ MEDICI L. Percutaneous treatment of pelvic congestion syndrome. Radiol Med 2003;105:76e82.
- COTRONEO AR, DI STASI C, TROPEANO G, SUMMARIA V, PEDICELLI A, CINA A. Percutaneous treatment of pelvic varicocele (abstract). Radiology 1998;209(Suppl):378e379.
- ECLAVEA A, YEAGER TD, CORDTS PR, VENBRUX A, BUCKLEY BJ. Pelvic congestion syndrome: multicenter results of embolotherapy (abstract). J Vasc Interv Radiol 1998;9(Suppl):184e185.
- MACHAN L, MOWATT J, HURWITZ T, DOYLE L, FRY P. Clinical outcome of women with chronic pelvic pain treated by ovarian vein embolization (abstract). J Vasc Interv Radiol 1998;9(Suppl): 185.
- TARAZOV PG, PROZOROVSKIJ KV, RYZHKOV VK. Pelvic pain syndrome caused by ovarian varices. Treatment by transcatheter embolization. Acta Radiol 1997;38:1023e1025.
- SICHLAU MJ, YAO JST, VOGELZANG RL. Transcatheter embolotherapy or the treatment of pelvic congestion syndrome. Obstet Gynecol 1994;83:892e896.
Table 1: Distribution of varicose veins in the group of 24 female patients presenting with pelvic venous incompetence. No sapheno-femoral junction were incompetent. Several types of varicose veins could be associated in a given patient
| Type of varices | N=24 |
|---|---|
| Perineal veins | 24 |
| Bilateral perineal veins | 11 |
| Right perineal veins | 5 |
| Left perineal veins | 8 |
| Vein of the sciatic nerve or fibular nerve | 2 |
| Perineal veins + great saphenous trunk | 9 |
| Recurrence after GSV stripping | 10 |
| Thigh or buttock perforators | 2 |
Table 2: Number of embolisations of corresponding incompetent pelvic veins
| Type of incompetent pelvic vein | Number of cases |
|---|---|
| Left ovarian vein | 11 |
| Left ovarian vein + left internal iliac vein | 5 |
| Left ovarian vein + right internal iliac vein | 2 |
| Right internal iliac vein | 3 |
| Left internal iliac vein | 2 |
| Left and right internal iliac vein | 1 |
Table 3: Long-term results in different series according to the % of patients experiencing improvement and % of clinical improvement at follow-up. ND = no data
| N | % of patients with improvement | % of clinical improvement | Follow-up months | ||
|---|---|---|---|---|---|
| Creton[current study] | 2006 | 24 | 50 | 100 | 36 |
| Pieri19 | 2003 | 33 | 61 | 100 | 9 |
| Garret17 | 2002 | 13 | ND | ND | 18 |
| Maleux16 | 2000 | 41 | 58.5 | 100 | 19.9 |
| Venbrux18 | 2000 | 56 | 65 | 100 | 12 |
| Cordt8 | 1998 | 11 | 40 | 100 | 13.4 |
| Cotroneo20 | 1988 | 22 | 60 | 100 | 3 |
| Eclavea21 | 1998 | 14 | 80 | ND | 12 |
| Machan22 | 1998 | 23 | 78 | ND | 15 |
| Tazarof23 | 1997 | 6 | 100 | 100 | 24 |
| Capasso9 | 1997 | 19 | 58 | 100 | 15.4 |
| Sichau24 | 1993 | 3 | ND | ND | 18 |
Fig. 1
Evolution of pelvic pain, venous pain and dyspareunia as assessed by VAS. The bar graph shows the mean scores for each symptom at the five observation points. The possible range of scores was 0 (no pain) e 10 (most severe pain).
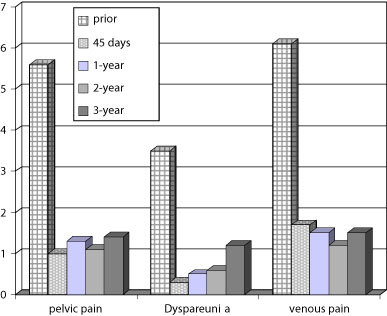
Fig. 2
Evolution of clinical scores for each patient prior to embolisation and up to 3-years follow-up. The vertical axis shows the global symptom score, obtained by summing each of the three symptoms scores at each time point. A maximum value of 30 is possible. One patient with only a 31% symptom improvement was considered to be a treatment failure.
| Date | 2007 |
| Auteurs | Eur J Vasc Endovasc Surg 2007;34:112-7 D.Creton, L. Hennequin, F. Kohler and F. Allaert |






